Abstract
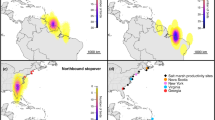
Arctic Terns (Sterna paradisaea) share a few routes to undertake the longest annual migrations of any organism. To understand how the wide spatial range of their breeding colonies may affect their migration strategies (e.g., departure date), we tracked 53 terns from five North American colonies distributed across 30° of latitude and 90° of longitude. While birds from all colonies arrived in Antarctic waters at a similar time, terns nesting in the Arctic colonies migrated back north more slowly and arrived to their breeding grounds later than those nesting in the colony farther south. Arrival dates in Antarctic waters coincided with the start of favorable foraging conditions (i.e., increased ocean productivity), and similarly arrival dates at breeding colonies coincided with the start of local favorable breeding conditions (i.e., disappearance of snow and ice). Larger birds followed a more direct southbound migration route than smaller birds. On both southbound and northbound migrations, daily distances traveled declined as time spent in contact with the ocean increased, suggesting a trade-off between resting/foraging and traveling. There was more unexplained variation in behavior among individuals than among colonies, and one individual had a distinctive stop around Brazil. Terns nesting in the Arctic have a narrow time window for breeding that will likely increase with continuing declines in sea ice and snow. Departing Arctic Terns likely have few clues about the environmental conditions they will encounter on arrival, and their response to environmental changes at both poles may be assisted by large individual variation in migration strategy.







Similar content being viewed by others
Data availability
The movement data are available on Movebank (Movebank ID: 727170503).
Code availability
The analyses used R packages available on CRAN or Github.
References
Alerstam T, Bäckman J, Grönroos J et al (2019) Hypotheses and tracking results about the longest migration: the case of the arctic tern. Ecol Evol 9:9511–9531. https://doi.org/10.1002/ece3.5459
Anderson AM, Duijns S, Smith PA et al (2019) Migration distance and body condition influence shorebird migration strategies and stopover decisions during southbound migration. Front Ecol Evol 7:1–14. https://doi.org/10.3389/fevo.2019.00251
Anderson CM, Gilchrist HG, Ronconi RA et al (2020) Both short and long distance migrants use energy-minimizing migration strategies in North American herring gulls. Mov Ecol 8:1–12. https://doi.org/10.1186/s40462-020-00207-9
Baak JE, Provencher JF, Mallory ML (2020) Changes in organ size and nutrient reserves of arctic terns (Sterna paradisaea) breeding near a High Arctic polynya. Arctic Antarct Alp Res 52:596–604. https://doi.org/10.1080/15230430.2020.1827577
Bartoń K (2020) MuMIn: multi-model inference. R package version 1.43.17. https://CRAN.R-project.org/package=MuMIn. Accessed 26 May 2021
Bêty J, Giroux JF, Gauthier G (2004) Individual variation in timing of migration: causes and reproductive consequences in greater snow geese (Anser caerulescens atlanticus). Behav Ecol Sociobiol 57:1–8. https://doi.org/10.1007/s00265-004-0840-3
Bogdanova MI, Butler A, Wanless S et al (2017) Multi-colony tracking reveals spatio-temporal variation in carry-over effects between breeding success and winter movements in a pelagic seabird. Mar Ecol Prog Ser 578:167–181. https://doi.org/10.3354/meps12096
Catry P, Dias MP, Phillips RA, Granadeiro JP (2013) Carry-over effects from breeding modulate the annual cycle of a long-distance migrant: an experimental demonstration. Ecology 94:1230–1235. https://doi.org/10.1890/12-2177.1
Comiso JC, Parkinson CL, Gersten R, Stock L (2008) Accelerated decline in the Arctic sea ice cover. Geophys Res Lett 35:1–6. https://doi.org/10.1029/2007GL031972
Conklin JR, Battley PF, Potter MA, Fox JW (2010) Breeding latitude drives individual schedules in a trans-hemispheric migrant bird. Nat Commun 1:1–6. https://doi.org/10.1038/ncomms1072
Corbeau A, Prudor A, Kato A, Weimerskirch H (2020) Development of flight and foraging behaviour in a juvenile seabird with extreme soaring capacities. J Anim Ecol 89:20–28. https://doi.org/10.1111/1365-2656.13121
Davies TE, Carneiro APB, Tarzia M et al (2021) Multi-species tracking reveals a major seabird hotspot in the North Atlantic. Conserv Lett 14(5):e12824
Devlin CM, Diamond AW, Saunders GW (2004) Sexing Arctic Terns in the field and laboratory. Waterbirds 27:314–320. https://doi.org/10.1675/1524-4695(2004)027[0314:SATITF]2.0.CO;2
Dias RA, Agne CE, Barcelos-Silveira A, Bugoni L (2012) New records and a review of the distribution of the Arctic Tern Sterna paradisaea Pontoppidan, 1763 (Aves: Sternidae) in Brazil. Check List 8:563–567. https://doi.org/10.15560/8.3.563
Dormann CF, Elith J, Bacher S et al (2013) Collinearity: a review of methods to deal with it and a simulation study evaluating their performance. Ecography (Cop) 36:27–46. https://doi.org/10.1111/j.1600-0587.2012.07348.x
Ducklow H, Carlson C, Church M et al (2001) The seasonal development of the bacterioplankton bloom in the Ross Sea, Antarctica, 1994–1997. Deep Res II Top Stud Oceanogr 48:4199–4221. https://doi.org/10.1016/S0967-0645(01)00086-8
Duffy DC, McKnight A, Irons DB (2013) Trans-Andean passage of migrating Arctic Terns over Patagonia. Mar Ornithol 41:155–159
Egevang C, Frederiksen M (2011) Fluctuating breeding of Arctic Terns (Sterna paradisaea) in Arctic and high-Arctic colonies in Greenland. Waterbirds 34:107–111. https://doi.org/10.1675/063.034.0114
Egevang C, Stenhouse IJ, Phillips RA et al (2010) Tracking of Arctic Terns Sterna paradisaea reveals longest animal migration. Proc Natl Acad Sci USA 107:2078–2081. https://doi.org/10.1073/pnas.0909493107
Fifield DA, Montevecchi WA, Garthe S, et al (2014) Migratory tactics and wintering areas of Northern Gannets (Morus bassanus) breeding in North America. In: Ornithological monographs No. 79. The American Ornithologists’ Union, pp 1–63
Fijn RC, Hiemstra D, Phillips RA, van der Winden J (2013) Arctic Terns Sterna paradisaea from the Netherlands migrate record distances across three oceans to Wilkes Land, East Antarctica. Ardea 101:3–12. https://doi.org/10.5253/078.101.0102
Hahn S, Emmenegger T, Lisovski S et al (2014) Variable detours in long-distance migration across ecological barriers and their relation to habitat availability at ground. Ecol Evol 4:4150–4160. https://doi.org/10.1002/ece3.1279
Hatch JJ, Gochfeld M, Burger J, Garcia E (2020) Arctic Tern (Sterna paradisaea). In: Billerman SM (ed) Birds of the world. Cornell Lab of Ornithology, Ithaca
Hijmans RJ, Williams E, Vennes C (2021) geosphere: spherical trigonometry. R package version 1.5–14. https://CRAN.R-project.org/package=geosphere. Accessed 14 Oct 2021
Holbech LH, Gbogbo F, Aikins TK (2018) Abundance and prey capture success of Common Terns (Sterna hirundo) and Pied Kingfishers (Ceryle rudis) in relation to water clarity in south-east coastal Ghana. Avian Res 9:1–13. https://doi.org/10.1186/s40657-018-0116-7
Horton KG, Van Doren BM, Stepanian PM et al (2016) Seasonal differences in landbird migration strategies. Auk 133:761–769. https://doi.org/10.1642/AUK-16-105.1
Hromádková T, Pavel V, Flousek J, Briedis M (2020) Seasonally specific responses to wind patterns and ocean productivity facilitate the longest animal migration on earth. Mar Ecol Prog Ser 638:1–12. https://doi.org/10.3354/meps13274
Levermann N, Tøttrup AP (2007) Predator effect and behavioral patterns in Arctic Terns (Sterna paradisaea) and Sabine’s Gulls (Xema sabini) during a failed breeding year. Waterbirds 30:417–420. https://doi.org/10.1675/1524-4695(2007)030[0417:PEABPI]2.0.CO;2
Liechti F, Bauer S, Dhanjal-Adams KL et al (2018) Miniaturized multi-sensor loggers provide new insight into year-round flight behaviour of small trans-Sahara avian migrants. Mov Ecol 6:19. https://doi.org/10.1186/s40462-018-0137-1
Lisovski S, Hahn S (2012) GeoLight—processing and analysing light-based geolocator data in R. Methods Ecol Evol 3:1055–1059. https://doi.org/10.1111/j.2041-210X.2012.00248.x
Lisovski S, Bauer S, Briedis M et al (2020) Light-level geolocator analyses: a user’s guide. J Anim Ecol 89:221–236. https://doi.org/10.1111/1365-2656.13036
Maftei M, Davis SE, Mallory ML (2015) Assessing regional populations of ground-nesting marine birds in the Canadian High Arctic. Polar Res 34:1–9. https://doi.org/10.3402/polar.v34.25055
Mallory ML, Forbes MR (2007) Does sea ice constrain the breeding schedules of High Arctic Northern Fulmars? Condor 109:894–906. https://doi.org/10.1093/condor/109.4.894
Mallory ML, Boadway KA, Davis SE et al (2017) Breeding biology of Arctic Terns (Sterna paradisaea) in the Canadian High Arctic. Polar Biol 40:1515–1525. https://doi.org/10.1007/s00300-016-2072-1
McKinnon L, Smith PA, Nol E et al (2010) Lower predation risk for migratory birds at high latitudes. Science 327:326–327. https://doi.org/10.1126/science.1183010
McKnight A, Allyn AJ, Duffy DC, Irons DB (2013) “Stepping stone” pattern in Pacific Arctic Tern migration reveals the importance of upwelling areas. Mar Ecol Prog Ser 491:253–264. https://doi.org/10.3354/meps10469
Moe B, Stempniewicz L, Jakubas D et al (2009) Climate change and phenological responses of two seabird species breeding in the high-Arctic. Mar Ecol Prog Ser 393:235–246. https://doi.org/10.3354/meps08222
Morten JM, Burgos JM, Collins L et al (2022) Foraging behaviours of breeding Arctic Terns Sterna paradisaea and the impact of local weather and fisheries. Front Mar Sci 8:760670. https://doi.org/10.3389/FMARS.2021.760670/BIBTEX
Neufeld LR, Muthukumarana S, Fischer JD et al (2021) Breeding latitude is associated with the timing of nesting and migration around the annual calendar among Purple Martin (Progne subis) populations. J Ornithol. https://doi.org/10.1007/s10336-021-01894-w
Nilsson C, Klaassen RHG, Alerstam T (2013) Differences in speed and duration of bird migration between spring and autumn. Am Nat 181:837–845. https://doi.org/10.1086/670335
Olmos F (2002) Non-breeding seabirds in Brazil: a review of band recoveries. Braz J Ornithol 10:31–42
Park J, Kuzminov FI, Bailleul B et al (2017) Light availability rather than Fe controls the magnitude of massive phytoplankton bloom in the Amundsen Sea polynyas, Antarctica. Limnol Oceanogr 62:2260–2276. https://doi.org/10.1002/lno.10565
Phillips RA, Lewis S, González-Solís J, Daunt F (2017) Causes and consequences of individual variability and specialization in foraging and migration strategies of seabirds. Mar Ecol Prog Ser 578:117–150. https://doi.org/10.3354/meps12217
Pinheiro J, Bates D, DebRoy S, et al (2021) nlme: linear and nonlinear mixed effects models. R package version 3.1–153. https://CRAN.R-project.org/package=nlme. Accessed 1 Nov 2021
Pratte I, Boadway KA, Davis SE et al (2017) Diet dichotomy between two migrant seabirds breeding near a high Arctic polynya. R Soc Open Sci. https://doi.org/10.1098/rsos.160982
Ramos R, Sanz V, Militão T et al (2015) Leapfrog migration and habitat preferences of a small oceanic seabird, Bulwer’s petrel (Bulweria bulwerii). J Biogeogr 42:1651–1664. https://doi.org/10.1111/jbi.12541
Redfern CPF, Bevan RM (2020) Overland movement and migration phenology in relation to breeding of Arctic Terns Sterna paradisaea. Ibis (Lond 1859) 162:373–380. https://doi.org/10.1111/ibi.12723
Reynolds RW, Smith TM, Liu C et al (2007) Daily high-resolution-blended analyses for sea surface temperature. J Clim 20:5473–5496. https://doi.org/10.1175/2007JCLI1824.1
Saba VS, Griffies SM, Anderson WG et al (2015) Enhanced warming of the Northwest Atlantic Ocean under climate change. J Geophys Res Ocean 121:118–132. https://doi.org/10.1002/2015JC011346
Sauve D, Divoky G, Friesen VL (2019) Phenotypic plasticity or evolutionary change? An examination of the phenological response of an arctic seabird to climate change. Funct Ecol 33:2180–2190. https://doi.org/10.1111/1365-2435.13406
Schielzeth H, Dingemanse NJ, Nakagawa S et al (2020) Robustness of linear mixed-effects models to violations of distributional assumptions. Methods Ecol Evol 11:1141–1152. https://doi.org/10.1111/2041-210X.13434
Schmaljohann H (2018) Proximate mechanisms affecting seasonal differences in migration speed of avian species. Sci Rep 8:1–10. https://doi.org/10.1038/s41598-018-22421-7
Seward A, Taylor RC, Perrow MR et al (2021) Effect of GPS tagging on behaviour and marine distribution of breeding Arctic Terns Sterna paradisaea. Ibis 163:197–212. https://doi.org/10.1111/IBI.12849
Smith RJ, Moore FR (2005) Arrival timing and seasonal reproductive performance in a long-distance migratory landbird. Behav Ecol Sociobiol 57:231–239. https://doi.org/10.1007/s00265-004-0855-9
Smith WO Jr, Marra J, Hiscock MR, Barber RT (2000) The seasonal cycle of phytoplankton biomass and primary productivity in the Ross Sea, Antarctica. Deep Res II Top Stud Oceanogr 47:3119–3140. https://doi.org/10.1016/S0967-0645(00)00061-8
Smith PA, Gilchrist HG, Forbes MR et al (2010) Inter-annual variation in the breeding chronology of arctic shorebirds: effects of weather, snow melt and predators. J Avian Biol 41:292–304. https://doi.org/10.1111/j.1600-048X.2009.04815.x
Sumner MD, Wotherspoon SJ, Hindell MA (2009) Bayesian estimation of animal movement from archival and satellite tags. PLoS ONE 4:19–22. https://doi.org/10.1371/journal.pone.0007324
U.S. Fish & Wildlife Service (2013) Field notes entry—geo-locators reveal migratory pathway of Arctic Terns. Online published results. www.fws.gov/FieldNotes/regmap.cfm?arskey=32639. Accessed 12 Nov 2019
Wong J, Lisovski S, Alisauskas R et al (2021) Arctic Terns from circumpolar breeding colonies share common migratory routes. Mar Ecol Prog Ser. https://doi.org/10.3354/meps13779 (In Press)
Wotherspoon SJ, Sumner MD, Lisovski S (2016) TwGeos: basic data processing for light-level geolocation archival tags. R package version 0.0–1
Acknowledgements
We thank all field technicians for their support in the field and two anonymous reviewers for their comments. Support to purchase the tracking units was provided by the Smithsonian Migratory Bird Center’s Migratory Connectivity Project and funded by the ConocoPhillips Charitable Investments Global Signature Program. Environment and Climate Change Canada, Natural Resources Canada (Polar Continental Shelf Program), ArcticNet Networks of Centers of Excellence, ConocoPhillips Alaska, and the Central and Mississippi Flyway Councils provided funding and logistic support to teams deploying geolocators. Financial support to purchase the computing equipment of JBW and MAM was provided by the Canada Foundation for Innovation (John R. Evans Leaders program) and the B.C. Knowledge Development Fund Program. MAM and MLM were supported by the Canada Research Chairs Program and Natural Sciences and Engineering Research Council of Canada. JBW was supported by the Werner and Hildegard Hesse Fellowship in Ornithology and the Ocean Leaders Graduate Fellowship.
Funding
Support to purchase the tracking units was provided by the Smithsonian Migratory Bird Center’s Migratory Connectivity Project and funded by the ConocoPhillips Charitable Investments Global Signature Program. Environment and Climate Change Canada and Natural Resources Canada (Polar Continental Shelf Program), ConocoPhillips Alaska, and the Central and Mississippi Flyway Councils provided funding and logistic support to teams deploying geolocators. Financial support to purchase the computing equipment of JBW and MAM was provided by the Canada Foundation for Innovation (John R. Evans Leaders program) and the B.C. Knowledge Development Fund Program. MAM and MLM were supported by the Canada Research Chairs Program and Natural Sciences and Engineering Research Council of Canada. JBW was supported by the Werner and Hildegard Hesse Fellowship in Ornithology and the Ocean Leaders Graduate Fellowship.
Author information
Authors and Affiliations
Contributions
JBW, MLM, and MAM conceived and designed the research based on previously collected data. RTA, WE, ALH, DKK, MM, ANM, RAR, PAS, and MLM designed, planned, and conducted the original field research. JBW, SL, and MAM designed and performed the analyses. JBW, MLM, and MAM wrote the manuscript. All authors read and approved the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no financial or proprietary interests in any material discussed in this article.
Ethical approval
All research was conducted under approved animal care protocols (Acadia University ACC 04-17; National Zoological Park ACUC Protocols 15-20, 18-06) and scientific permits (e.g., Environment and Climate Change Canada (ECCC) Banding: 10694, Government of Nunavut Wildlife License 2018-038; US Federal Bird Banding Permit: 09700, Alaska Scientific Permits: 17-162, 18-156).
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Wong, J.B., Lisovski, S., Alisauskas, R.T. et al. Variation in migration behaviors used by Arctic Terns (Sterna paradisaea) breeding across a wide latitudinal gradient. Polar Biol 45, 909–922 (2022). https://doi.org/10.1007/s00300-022-03043-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00300-022-03043-2